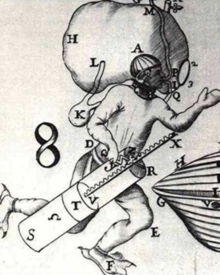
One of the earliest "rebreather" designs. In 1680, Giovanni Borelli envisioned a diver carrying a large bag of air from which the diver breathed, as necessary. Click image for larger view and image credit.
Technical Diving
Marc Slattery
Chief Scientist – Cayman Islands Twilight Zone 2007
Department of Pharmacognosy
University of Mississippi
Ocean exploration and research have advanced significantly since the 19th century, when most samples were obtained by dragging nets behind research vessels, or by hand-collecting shallow-water organisms from rocky shore pools during monthly low tides. By the mid-20th century, marine scientists were either conducting deep-sea research from ships using submersible technologies, or shallow-water research using self-contained underwater breathing apparatus (SCUBA) to dive. (The technology has become so prevalent that we no longer refer to it by its acronym, written in all capital letters, but instead refer to it as simply "scuba," a word onto itself.)
Each method has its strengths and weaknesses, but both have proven to be effective methods of exploring and accessing submerged habitats for a variety of research goals. In general, submersibles tend to be very expensive and they require highly trained operators to conduct work on behalf of the scientist. On the other hand, they can enter deep-water sites (thousands of meters beneath the surface) that scuba divers cannot access. In contrast, scuba is typically safety-limited to depths of less than 50 meters, but the costs are minimal and, with limited training, the individual scientist can actually access their own research samples. Unfortunately, the pragmatic need to focus limited and costly submersible time on deeper-water communities, and the safety limits of scuba, have meant that significant regions of the marine environment have been largely unexplored. In fact, we may know more about Mars than we do about the interface between shallow reefs and the deep sea.
The technical diving research team (shown here in the Bahamas in 2002) with assorted tanks carrying different mixtures of Trimix — a breathing gas consisting of oxygen, helium, and nitrogen. Click image for larger view and image credit.
A more recent version of the rebreather allows this diver to swim — without releasing bubbles — over a Caribbean reef. In addition to conserving air, this technique is particularly useful for marine biologists as fish behavior is not disturbed by the noise of exhaled bubbles. Click image for larger view and image credit.
Open circuit scuba has evolved over the last 150 years, and it has benefited from a quantum leap in the technology (namely, the Cousteau-Gagnon demand regulator) in the 1940s. Nonetheless, the principles of scuba have remained largely unchanged through time. A gas (typically air, but see below for other mixtures) is carried to depth within a solid container (the tank), and the diver accesses the gas in that container through some form of breathing tube/mouthpiece (the regulator). Over time, divers learned lessons, often painfully, regarding the safety limitations of scuba.
We now understand that breathing compressed air at depth has physiological consequences:
1. Pure oxygen is toxic at pressures consistent with a depth of 20 ft (not a problem for scuba divers who breathe typical atmospheric air — a mixture of 78% nitrogen, 21% oxygen, and 1% carbon dioxide);
2. Because of the physical properties of the constituents within a gas under pressure (i.e., Dalton’s Law of Partial Pressures), air becomes toxic at 218 ft due to its proportion of oxygen;
3. Nitrogen also has issues under pressure since gases are absorbed by the human body in direct proportion to their partial pressures (i.e., Henry’s Law), and the accumulation of significant nitrogen results in a narcotic effect at depths below 100 ft.
When nitrogen off-gassing occurs too rapidly on ascent, the bubbles trapped in tissues causes decompression sickness or “the bends.” In order to counteract these issues, technical divers blend gases to produce a mixture that is no longer similar to atmospheric air. The most common blends include Nitrox, an oxygen-enhanced/nitrogen-limited mixture that reduces decompression penalties, but it is typically not used deeper than 100 ft due to potential oxygen toxicity. Trimix is another blend that lowers the percentage of oxygen and nitrogen by adding another inert gas — helium. This mixture can be safely used beyond normal diving depths. (There can be additional consequences of scuba diving that can result in severe injuries, and death, so safety is a primary concern for both the recreational and the research diver.)
The term "open circuit" refers to the fact that gas is released into the surrounding water when the diver exhales into the regulator mouthpiece. In contrast, a "closed-circuit" system recycles the exhaled gas through a carbon dioxide “scrubber” that separates oxygen from carbon with each passage of gas. Thus, in theory, the diver carries unlimited gas since our body is able to utilize the recycled oxygen. (In reality, scrubber limitations result in reduced recycling efficiency with each passage of the gas through the breathing loop.) A closed-circuit rebreather (CCR) is also able to maintain a constant pO2, and thus an optimum pO2 as depth changes. This offsets the physiological limits imposed from inert gas uptake under pressure, such as decompression obligations and nitrogen narcosis. Therefore, rebreathers increase bottom-time (allowing more work), and they decrease decompression while increasing safety.
The Twilight Zone explorers will use open- and closed-circuit technical diving in the course of this project. Check the Ocean Explorer Web site, where our expedition logs will explain our dive profiles and the considerations that lead us to use one or the other technique.
Sign up for the Ocean Explorer E-mail Update List.





















