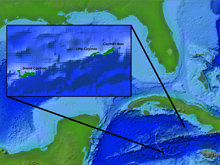
The Cayman Islands Twilight Zone 2007 expedition will take place in the Caribbean Sea, between Cuba and Jamaica. The image shows a view of the islands' rise from the abyss of the Cayman Trench, 7,500 meters (m) below the crystal-clear surface waters. Click image for larger view and image credit.
The zones profile of a typical coral reef. The deep fore-reef, or Twilight Zone, ranges from about 50- to 150-m depth and has rarely been explored. Click image for larger view and image credit.
Typical sponge biodiversity of a shallow Caribbean coral reef. Preliminary data from the co-principal investigators suggest that sponges on the deep reefs may be equally diverse. Click image for larger view and image credit.
Mission Plan
Marc Slattery
Chief Scientist — The Cayman Islands Twilight Zone 2007
Department of Pharmacognosy
University of Mississippi
![]() NOAA Podcast:
NOAA Podcast:
Learn what Marc Slattery hopes to discover on this expedition with a video or audio podcast on the mission.
Why study deep-reef communities?
Recent interest in deep-reef communities has been sparked by an increased understanding that they represent important refuges and nursery habitats for shallow-water coral and fish populations. Thus, as shallow-water reefs suffer declines, the deep reefs might be one of the few natural options for recovery.
Exploration Site: Little Cayman Wall, Cayman Islands
The Cayman Islands (19°41’ N, 80°03’ W) rise from the Cayman Trench, located in the center of the Caribbean Sea, between Cuba and Jamaica. Upwelling of deep, nutrient-rich waters from the North Equatorial and Caribbean currents aid in the development of shallow (2- to 20-meter depth) fringing reefs and patch reefs. Many of these reefs are associated with mangrove forests, sea grass beds, and algal plains, which are important habitat for reef-associated fishes.
The Cayman Island reefs are considered some of the most pristine within the Caribbean due to limited human populations and impacts. They provide an opportunity to study coral reef communities (shallow and deep), with a focus on natural processes. The reefs surrounding Little Cayman are significant for being the least impacted within the Cayman Islands, and for the narrow “skirt” of shallow fringing and patch reefs near shore, so the walls are often located within 0.5 kilometers (km) of the beach. They include a zone of shallow-reef species on near vertical slopes, followed by a deeper zone of unique deep-reef species of sponges and corals, as well as some gorgonians (sea fans and sea whips) and algae. Investigating these deep, light-limited, coral reef systems that include many species of hermatypic (reef-building) corals will allow us to evaluate the potential negative impacts of several factors that broadly influence the decline or recovery of Caribbean coral reef systems.
Exploration Goals
The principle objective of this project is to increase current understanding of the transition zone between shallow tropical coral reefs and the aphotic (lacking in light) deep-water communities. Prior exploration and discovery in this zone is limited. The few surveys conducted to date provide little information about the extent or distribution of the transition zones, and their biodiversity and ecology.
We have four primary objectives for our project:
- Gain access to and explore the deep fore-reef communities of Little Cayman, with emphasis on sites representing a variety of slope angles and current patterns.
- Characterize the biodiversity and distribution patterns of these communities.
- Assess the site-specific flora and fauna for potential biotechnological uses.
- Provide a mechanism to include environmental education and outreach opportunities for K-12 teachers and students.
In addition, the following secondary objectives will further our biological understanding of these unique deep-reef communities:
- Examine deep-reef coral biology, including reproductive status, photosynthetic capability, growth rate, etc.
- Analyze phenotypic plasticity relevant to chemical variability across sites and over the depth gradient.
- Assess the connectivity of the deep fore-reefs to shallow reefs as it relates to disease transmission, genetic relatedness/gene flow, heat tolerance, and coral physiology.
Methods
• Access and Exploration. A site approximately 1 km northeast of the Little Cayman Research Center has been identified as our primary research site for this project. We will work this site almost exclusively in the first year of this grant. This site was chosen for a number of reasons, including representativeness to nearby deep reefs and proximity to the NOAA CREWS buoy from which environmental data can be downloaded.
The proposed research will be conducted using open-circuit scuba (i.e., air, Nitrox, and Trimix protocols) and closed-circuit rebreather (CCR) diving. Over the past five years, we have assembled a group of about 10 divers who are trained in several mixed gas techniques, including four trained to use open-circuit Trimix down to a depth of 125 meters (m), and four that have completed Level 1 CCR as of May 2006. This deep technical diving is sanctioned by our respective Diving Control Boards and the American Academy of Underwater Sciences. We are poised to make significant progress in this project using our existing open circuit training, and we will quickly advance to subsequently deeper levels of CCR training to increase our bottom time and productivity.
• Biodiversity and Distributions. A SONY 1000 3 CCD and housing rated to 100 m (pressure tested by us to 150 m) with high intensity discharge (HID) lights and convergent laser light system (to maintain camera at 40 cm above the substrate being photographed) will be used for replicate video surveys. Digital images or captured digital video frames will be analyzed using image analysis software for the abundance of corals and sponges at each depth. The data from the transect studies will be used to quantify total species number, individual species abundances, and diversity using diversity indices which can then be compared statistically over the full bathymetric range. In addition to hard corals and sponges, relevant data on the community assemblages (from bacteria to fishes) can be used to address fundamental ecological relationships, and broad biodiversity comparisons across the regional reefs. These data will be archived in the Census of Marine Life project.
Measurements of the following will occur as water column profiles in Little Cayman during our time in the field and at other times when possible. Profiling will occur with a Seabird 19 CTD (conductivity-temperature-depth) with a calibrated fluorometer to measure chlorophyll a, and a light meter that measures photosynthetically active radiation (PAR: 400 to 700 nm). HOBO temps (Onset, Inc) will be set individually or in strings to measure temperature over long time periods of six months to one year at specific depths. Spectral irradiance, both visible and ultraviolet radiation (300 to 700 nm) will be measured using a LiCor LI-1800UW scanning spectroradiometer (LiCor, Lincoln, Nebraska) calibrated with National Institute of Standards and Technology (NIST) traceable standards. Scans will be taken at pre-determined depths and reported in units of W m-2 nm-1 at approximately 1300 hr daily. These data will be coordinated with the CREWS buoy that includes a meteorological station and oceanographic sensors (wind speed, wind direction, air temperature, barometric pressure, sea temperature, salinity, ultraviolet-B radiation) above and below the water, photosynthetically active radiation (PAR, above and below water), and tide (pressure as proxy).
• Biotechnology Potential. Minimal tissue from each sample collected for taxonomic purposes will be frozen at Little Cayman Research Center (LCRC), and transported back to the three academic institutions for biotechnological applications. The University of Mississippi (UM) will be responsible for extracting chemical compounds from the sponges and soft corals, and testing them in a variety of biomedical assays focused on new pharmaceuticals to prevent cancer, bacterial and fungal infections, and malaria. The University of New Hampshire will assess each sample for fluorescent properties of these species that can be developed into new molecular biology probes. The University of Alabama will isolate and culture the microbial communities associated with the deep-reef macroorganisms. They will then extract the culture media, and forward these chemical compounds to UM, where they will be tested in the aforementioned biomedical assays.
• Education and Outreach. A “teacher-in-residence" has been identified as part of the exploration team. Called an "Educator at Sea," she will work with the Ocean Explorer Education Office to develop lesson plans related to the expedition goals. She will also help relate the purpose of this expedition to a broader community through unique insights and experiences, as she works in the field and lab with the research team.
Related Links
National Institute for Undersea Science and Technology – Ocean Biotechnology Center and Repository
http://www.usm.edu/niust/obcrhome.htmNational Center for Natural Products Research
http://www.pharmacy.olemiss.edu/ncnpr/index.htmlLittle Cayman Research Center
http://www.reefresearch.org




















![The zones profile of a typical coral reef. The deep fore-reef, or Twilight Zone [about 50-150 m], has rarely been explored.](reef_diagram_220.jpg)
